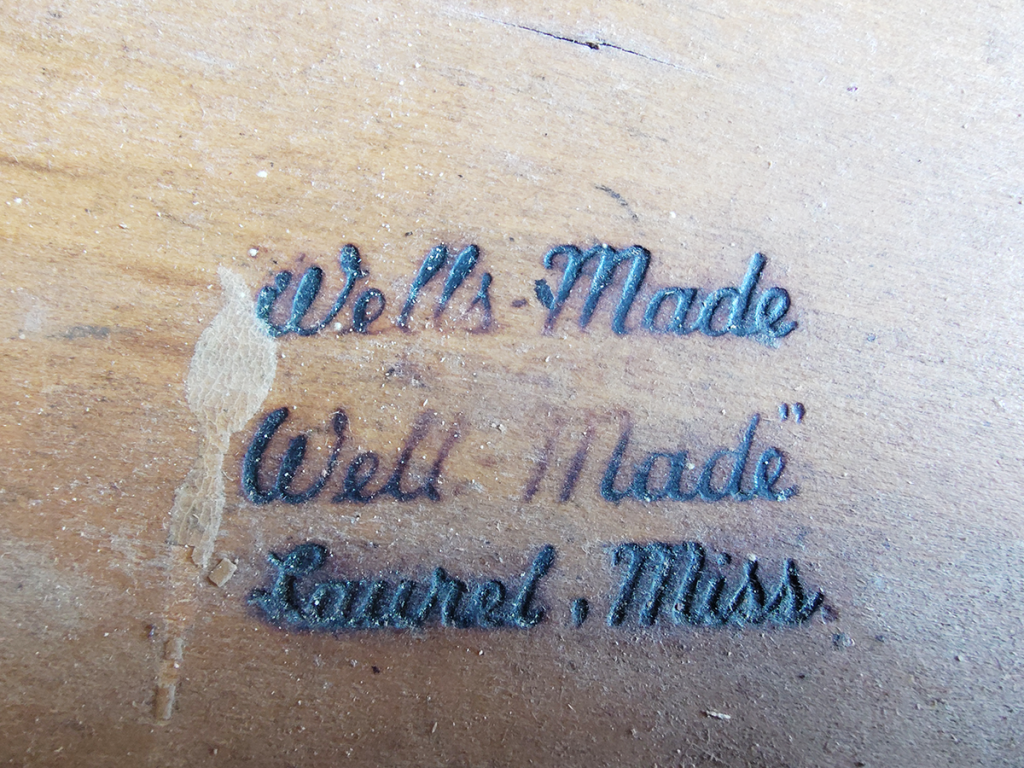
Discovered inside an old wooden desk, burnt into the solid-wood drawer. No complaints about the quality of construction, to be honest. But how long has it been since furniture makers burnt in their brand with red-hot iron?
Discoveries in the Physics & Astronomy shop | Science, curiosities, and surprises
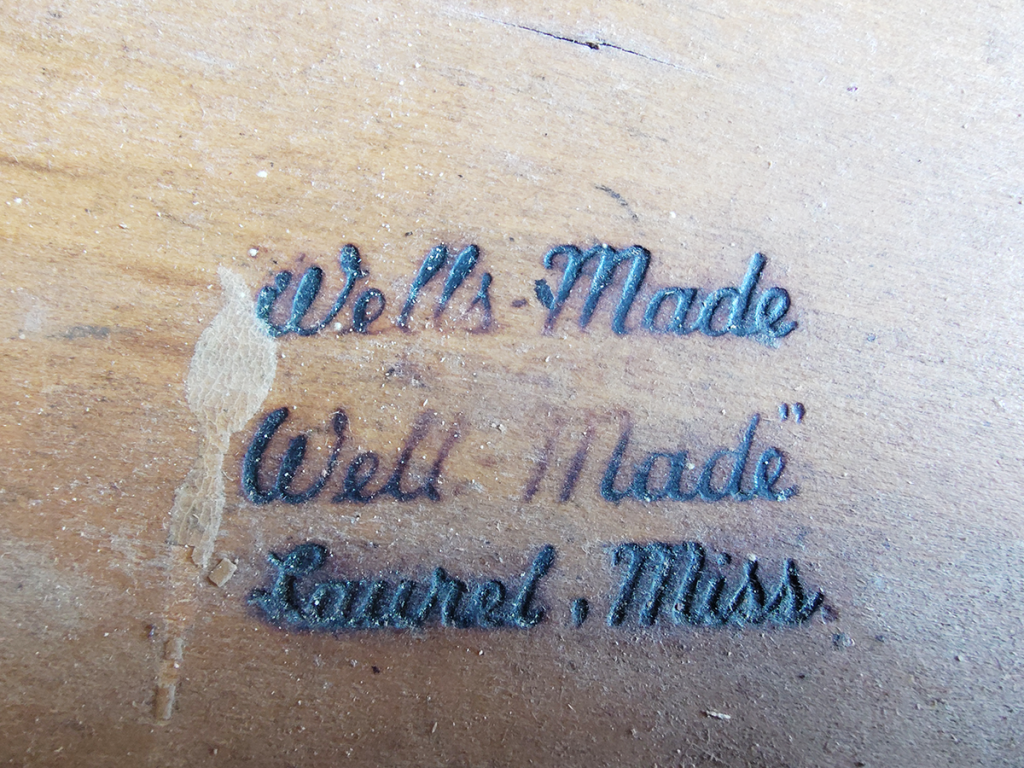
Discovered inside an old wooden desk, burnt into the solid-wood drawer. No complaints about the quality of construction, to be honest. But how long has it been since furniture makers burnt in their brand with red-hot iron?

Large and heavy coils of enamel-clad copper wire, which allow for tight, dense windings, are useful for demonstrating induced currents caused by a changing magnetic field. Students can spin magnets on a rubber band to produce enough electrical energy to light an LED. Not much, sure, but it’s still borderline magical.
Note that these hold up for a long time. Acquired in April of 1966 – just shy of six decades ago – and it’s still in regular use.
That $115.00 price marked on there? $1,171.89 in today’s dollars. Do you think we’ve recouped our investment?

“Classroom door key.” Matter-of-fact, handwritten on a torn adhesive label. No need for building or specific room number identification. (The other side has no markings.)
Maybe there was only one classroom at the time? Maybe they were all keyed identically? Maybe the original bearer was only concerned with one specific classroom, one which needed no elaboration? Who knows?

Another coffee can, this one half-filled with old keys, most unlabeled, almost certainly all of them to locks long gone. Not like “bucket of keys” is a prime organizational strategy, so one supposes these can be used for some exciting new arts and crafts project?

Apparently, at some indeterminate point in time, the second floor held cabinets with a breathing apparatus inside. Respirators? SCBA systems like firefighters use? SCUBA systems like divers use? Pretty confident that those’re long gone.
This one has become disconnected from its key, so your guess is as good as ours. What useful information could that possibly offer? Like, even if we knew which key it identified?

Sometimes you find oddities whose initial and continued existence boggles the mind. This clock was gathering dust atop the bookcases in the student lounge, battery-free and long-forgotten.
Where did it come from? What life did it live before it came to Olin? Who thought enough of it to acquire, but not enough to take with them?
What should we do with it now?

If it still works, it stays in service. There’s always a bit of “what’s this?” and “what’s it do?” and “how do I make it do what it does?” when stumbling across old equipment, but that’s what we have physicists for.

Sometimes you stumble across a delightful artifact. One with an unknown, perhaps unknowable history. Clearly, at one point, it was necessary to hold an object in a particular place, and none of the available clips, clamps, or clasps were up to the task.
A steel rod, an alligator clip, and some electrical tape to the rescue!
What’s fascinating about this isn’t the specifics of the object, but the way that these temporary, stopgap solutions can linger. After enough time and use, they become ordinary and unremarkable. Familiar.
Until, some indefinite period of years later, a fresh set of eyes spots them in an old drawer. Look at what’s in here!

These are not flashbulbs, merely incandescent A-lamps, but check out the sweet vintage ad!
So the “blue dot” thing was a quality indicator that Sylvania used in their classic flashbulbs. A magnesium filament inside an oxygen environment within the bulb produced a bright flash of light, assuming the envelope hadn’t been compromised. A dot of anhydrous cobalt (II) chloride inside that zero-moisture bulb would remain blue. If there was a leak, atmospheric moisture would react with the cobalt chloride to turn it pink, an indicator that this bulb might not work.
What’s less clear is how, exactly, one wee flashbulb is going to effectively illuminate that whale – flash photography being not super effective at distance – but whatever.

Fifty-five years ago saw the launch of the Apollo 13 lunar mission, on 11 April 1970. It didn’t go to plan, of course. Things took a bad turn, could have been worse, but who can blame them for optimism in the weeks leading up to it all? Moon science is cool!

Maps, plans, a pretty tight itinerary. It’s expensive and difficult to go to the Moon, so you don’t waste time. But don’t those hand-drawn maps just make it so inviting? Presumably the astronauts carried maps which were much more detailed and useful, if less likely to get the kid inside all of us super-excited.
Apollo 14 would reach the Fra Mauro highlands in early 1971, though Lovell, Swigert, and Haise never flew into space again.

Here, our more modern sodium light sources, using a clever design that enabled a reduction from the minimum 35W to as little as 18W in 1977. Cool, yeah?
Unfortunately, Philips finally bowed out of the low pressure sodium lamp game in 2019, mean we’ve got these dinosaurs running for as long as we can scour spare lamps online. Once the supply’s gone, it’s gone.

Hey, look! Sodium metal! Highly reactive, so it’s inside with a mixture of 99% neon and 1% argon, neither of which deigns to react with, well, anything. That’s why, when it starts up, we see a purplish glow from the noble gases before tube reaches 260°C and vaporizes the sodium. After that, it’s an intense monochromatic yellow-orange that’s hard to look at.